Ocrevus Health Canada
Early PPMS will be defined by duration of disease and level of disability measured using Expanded Disability Status Scale EDSS. This website is neither intended nor designed to record or report adverse event information.
It is based on marijuana and ocrelizumab the active ingredients of Cannabis and Ocrevus respectively and Cannabis and Ocrevus the brand names.
Ocrevus health canada. Health Canada Approves OCREVUS ocrelizumab for 9 mins readOCREVUS is a new treatment for a rare and worsening form of MS that impacts 10-15 per cent of Canadians living with the disease 1 after negotiations between NHS England and Roche4It is the first approved treatment for the primary progressive form of MS 2483 for a supply of 10 milliliters called CD20000 to more than. Ocrevus was approved with conditions by Health Canada as monotherapy for the management of adult patients with early primary progressive multiple sclerosis PPMS as defined by disease duration and level of disability in conjunction with imaging features characteristic of inflammatory activity. Ocrelizumab acts as an immunomodulatory drug by targeting and removing potentially harmful B cells a type of.
It is not known if. Primary progressive MS in adults. This policy only applies to Health Partners Plans HPP Medicare product lines.
Canadian Agency for Drugs and Technologies in Health. Health Canadas approved Ocrevus ocrelizumab to treat adults with relapsing-remitting MS a move welcomed by that nations MS Society and others. You can also report adverse events relating to any product to Health.
Approved Drug Products containing Ocrevus listed with Health Canada. Generic drugs are not considered. OCREVUS is a new treatment for a rare and worsening form of MS that impacts 10-15 per cent of Canadians living with the disease1MISSISSAUGA ON F.
Hoffmann-La Roche Limited 7070 Mississauga Road Mississauga Ontario L5N 5M8. A Montreal man is going public with his fight to urge Health Canada to approve a promising new drug to help treat multiple sclerosis. MISSISSAUGA ON Feb.
The study uses data from the FDA. Ocrevus 19955 reports How the study uses the data. The federal agency Health Canada approved Ocrevus ocrelizumab for the treatment of adults with relapsing-remitting multiple sclerosis RRMS with.
Health Canada Approves OCREVUS ocrelizumab for Canadians Living with Relapsing-Remitting Multiple Sclerosis RRMS Canada NewsWire MISSISSAUGA ON Aug. 15 2017 OCREVUS is a first-in-class. OCREVUS is a prescription medicine used to treat.
Roche Genentech s Ocrevus can be prescribed to adults with early-stage PPMS and characteristic signs of inflammatory activity. Other drugs that have the same active ingredients eg. 15 2018 CNW - Hoffmann-La Roche Limited Roche Canada today announced that Health Canada has approved OCREVUS ocrelizumab with conditions as a monotherapy for the.
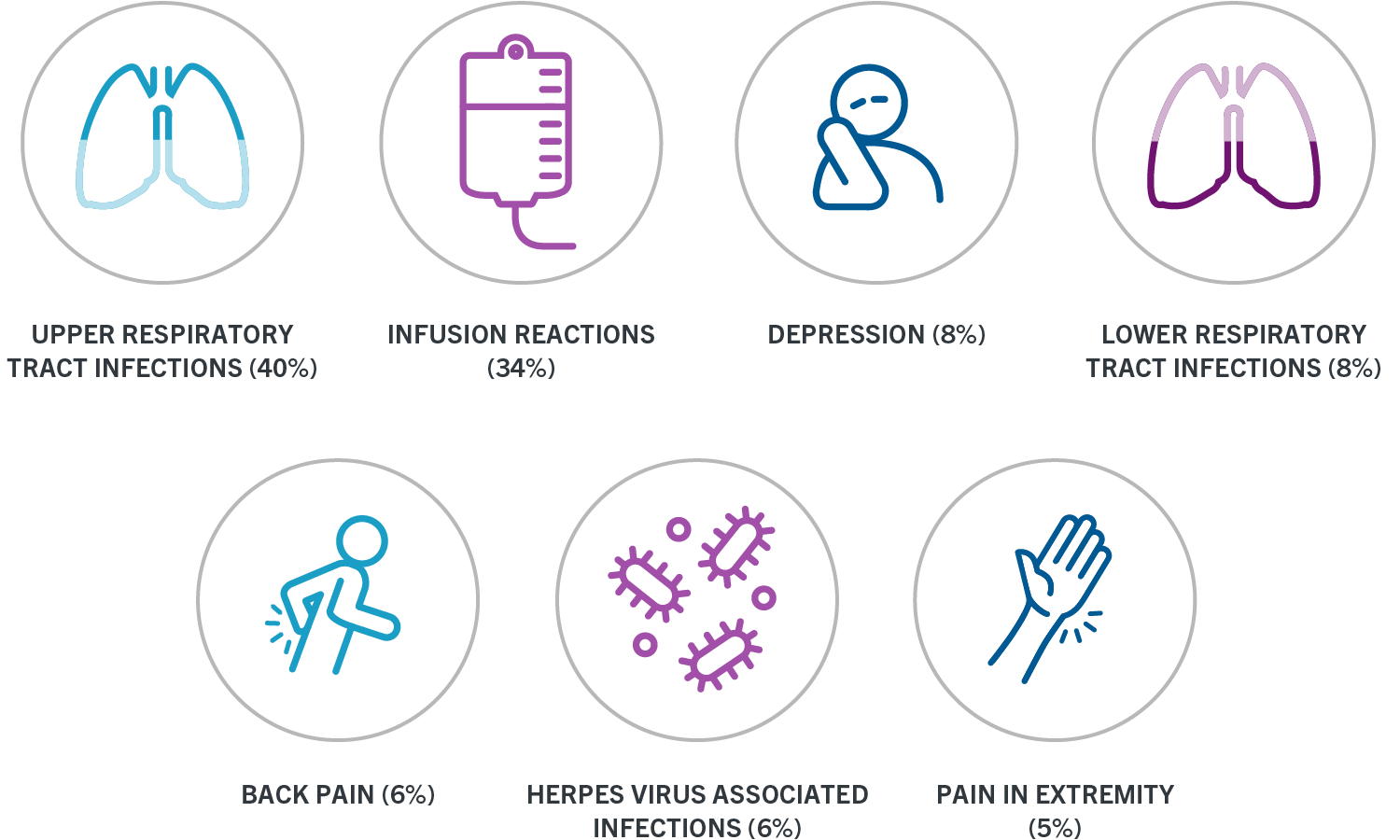
Delay OCREVUS administration in patients with an active infection until the infection is resolved. Early PPMS will be defined by duration of disease and level of disability measured using Expanded Disability Status Scale EDSS. A higher proportion of OCREVUS-treated patients experienced infections compared to patients taking IFN.
FDA APPROVED INDICATIONS Ocrelizumab is a CD20-directed cytolytic antibody used to treat patients with relapsing or primary progressive forms of multiple sclerosis. Health Canada approved Ocrevus ocrelizumab with conditions for the treatment of adults with early primary progressive MS PPMS. Roche Canada announced the approval of Ocrevus ocrelizumab with conditions for the treatment of people with early primary progressive MS PPMS.
As Globals Anne Leclair reports Health Canada. Management of adult patients with early primary progressive multiple sclerosis as defined by disease duration and level of disability in conjunction with imaging features characteristic of inflammatory activity Internet. Treatment with OCREVUS ocrelizumab should be initiated and supervised by neurologists experienced in the treatment of patients with MS and who have fully familiarized themselves with the efficacy and safety profile of OCREVUS.
In RRMS clinical trials 58 of OCREVUS-treated patients experienced one or more infections compared to 52 of IFN-treated patients. In Canada as elsewhere it is. Mississauga ON L5N 5M8.
Relapsing forms of multiple sclerosis MS to include clinically isolated syndrome relapsing-remitting disease and active secondary progressive disease in adults.
Https Mssociety Ca Library Document N6sungtrc5flmxpx4oeobqjcieftdgm2 Original Pdf
Https Www Cadth Ca Sites Default Files Cdr Clinical Sr0542 Ocrevus Ppms Cl Report Pdf
Ocrevus Added To Nhs For Rrms Patients In England And Wales But Not In Scotland
Buy Ocrevus Ocrelizumab Online Price Costs Thesocialmedwork
Health Canada Approval Of Multiple Sclerosis Drug Absolutely A Game Changer Globalnews Ca
Montrealer With Ms Calling On Health Canada To Approve Breakthrough Drug Ocrevus Montreal Globalnews Ca
Roche Pan Canadian Pharmaceutical Alliance Wrap Ocrevus Negotiations
Pharmacoeconomic Review Report Ocrelizumab Ocrevus Ncbi Bookshelf
Https Www Cadth Ca Sites Default Files Cdr Complete Sr0542 Cdr Complete Ocrevus Ppms Apr 30 18 E Pdf
Defining Ocrelizumab S Role Within The Ms Treatment Landscape Approvals Resources Thesocialmedwork
Https Www Cadth Ca Sites Default Files Cdr Complete Sr0519 Ocrevus Rms Complete Nov 23 17 Pdf
Multiple Sclerosis Subtypes Symptoms Diagnosis And Treatments Disabled World
Clinical Review Report Ocrelizumab Ocrevus Ncbi Bookshelf
Cadth Canadian Drug Expert Committee Recommendation Ocrelizumab Ocrevus Hoffmann La Roche Limited Ncbi Bookshelf
After Ocrevus Approval New Ms Therapies May Become Available
Pdf Ocrelizumab Induced Alopecia Areata A Series Of Five Patients From Ontario Canada A Case Report
Https Pdf Hres Ca Dpd Pm 00050590 Pdf




Posting Komentar untuk "Ocrevus Health Canada"